Naveed A
Level 2
ISO, Regulatory Affairs FDA, EU, UK, PMN 510k, QMS and Professionalism
Skills

Bekijk mijn diensten

Wil je op uurbasis werken?
Vertel Naveed A wat je nodig hebt.
US$ 100
/
uurPortfolio
Werkervaring
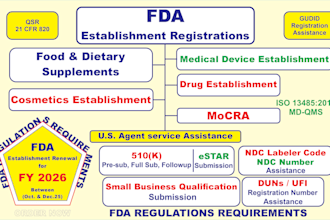
Senior Regulatory Affairs & QMS Consultant | EU MDR, FDA 510(k), ISO 13485
MDR Advisory • Freelance
Oct 2022 - Present • 3 yrs 7 mos
Regulatory Affairs & QMS Consultant with 25+ years of experience supporting clients across medical devices, IVDs, cosmetics, food, and pharmaceuticals. Specialized in EU MDR, UKCA, and FDA compliance, including 510(k) submissions and FDA Establishment Registration. Key achievements include preparation of Technical Documentation/Technical Files, development of ISO 13485 QMS documentation, and execution of risk management (ISO 14971), CER, and IFU compliance. Conducted gap assessments, internal audits, and provided practical regulatory strategies to ensure audit readiness and efficient market access. Focused on delivering clear, compliant, and actionable solutions tailored to client needs.
91 Reviews
| (86) | ||
| (2) | ||
| (1) | ||
| (0) | ||
| (2) |
Specificering van de beoordeling
- Communicatieniveau van de freelancer
- Kwaliteit van de levering
- Waarde van de levering
Sorteer op
dolivierp
Terugkerende klant

Argentinië

ilanohayon443
Terugkerende klant

Verenigde Staten

lukehunter318
Terugkerende klant

Verenigde Staten
My 3rd year in a row working with him, always a pleasure.
Reactie van de freelancer
jasonwrightevo
Terugkerende klant

Verenigd Koninkrijk
Reactie van de freelancer
jasonwrightevo
Terugkerende klant

Verenigd Koninkrijk